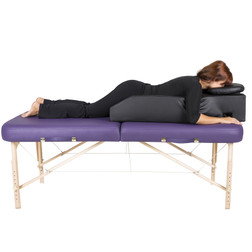
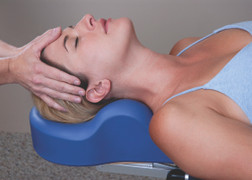
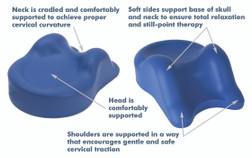
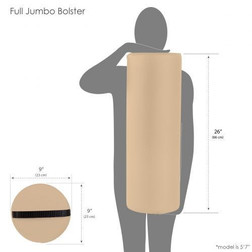
All Products
-
$35.99
-
$374.99
-
$45.99
-
MSRP: $79.00$69.00
-
$32.99
-
$35.99
-
$39.99
-
$69.00
-
$29.99
-
$110.00
-
$120.99
-
$59.99
-
$59.00 - $90.00
-
$43.99
-
$34.99